8.How to Read a Steam Table
Saturated Steam Tables
A saturated steam table is an indispensable tool for any engineer working with steam. It's typically used to determine saturated steam temperature from steam pressure, or the opposite: pressure from saturated steam temperature. In addition to pressure and temperature, these tables usually include other related values such as specific enthalpy (h) and specific volume (v).
The data found in a saturated steam table always refers to steam at a particular saturation point, also known as the boiling point. This is the point where water (liquid) and steam (gas) can coexist at the same temperature and pressure. Because H2O can be either liquid or gas at its saturation point, two sets of data are required: data for saturated water (liquid), which is typically marked with an "f" in subscript, and data for saturated steam (gas), which is typically marked using a "g" in subscript.
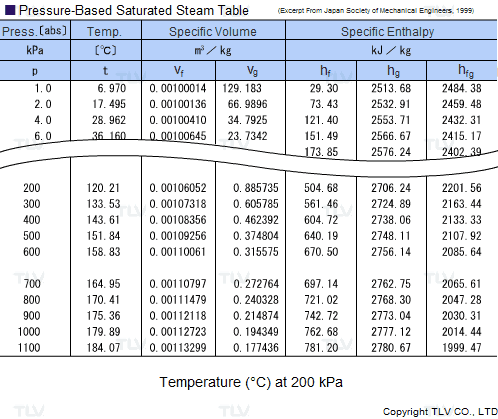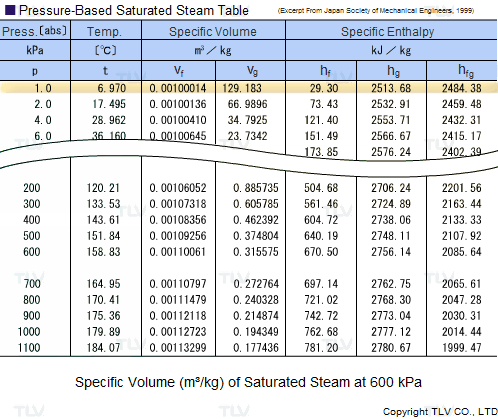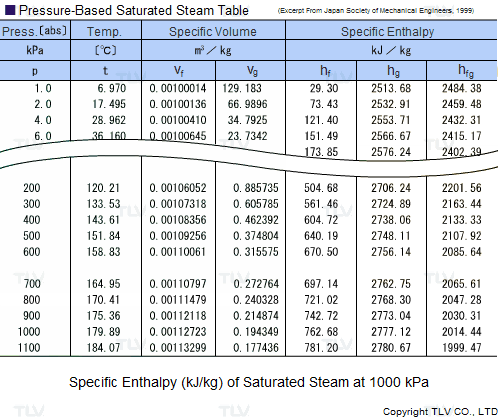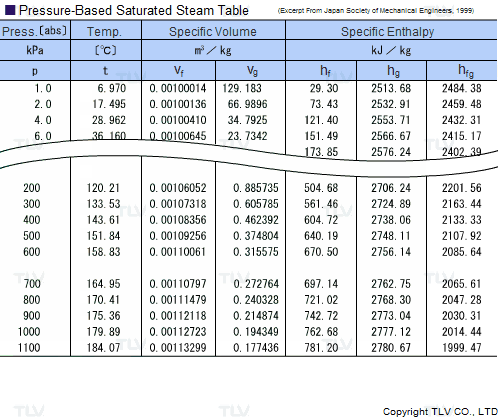
| Example of Saturated Steam Table |
|---|
|
|
|
Legend:
|
Heating processes using steam generally use the latent heat of evaporation (Hfg) to heat the product. As seen in the table, this latent heat of evaporation is greatest at lower pressures. As saturated steam pressure rises, the latent heat of evaporation gradually decreases until it reaches 0 at supercritical pressure, i.e. 22.06 MPa (3200 psi).
Two Formats: Pressure Based and Temperature Based
Since saturated steam pressure and saturated steam temperature are directly related to one another, saturated steam tables are generally available in two different formats: based on pressure and based on temperature. Both types contain the same data that is simply sorted differently.
Pressure Based Saturated Steam Table
| Press. (Gauge) |
Temp. | Specific Volume | Specific Enthalpy | |||
|---|---|---|---|---|---|---|
| kPaG | °C | m3/kg | kJ/kg | |||
| P | T | Vf | Vg | Hf | Hfg | Hg |
| 0 | 99.97 | 0.0010434 | 1.673 | 419.0 | 2257 | 2676 |
| 20 | 105.10 | 0.0010475 | 1.414 | 440.6 | 2243 | 2684 |
| 50 | 111.61 | 0.0010529 | 1.150 | 468.2 | 2225 | 2694 |
| 100 | 120.42 | 0.0010607 | 0.8803 | 505.6 | 2201 | 2707 |
Temperature Based Saturated Steam Table
| Temp. | Press. (Gauge) |
Specific Volume | Specific Enthalpy | |||
|---|---|---|---|---|---|---|
| °C | kPaG | m3/kg | kJ/kg | |||
| T | P | Vf | Vg | Hf | Hfg | Hg |
| 100 | 0.093 | 0.0010435 | 1.672 | 419.1 | 2256 | 2676 |
| 110 | 42.051 | 0.0010516 | 1.209 | 461.4 | 2230 | 2691 |
| 120 | 97.340 | 0.0010603 | 0.8913 | 503.8 | 2202 | 2706 |
| 130 | 168.93 | 0.0010697 | 0.6681 | 546.4 | 2174 | 2720 |
| 140 | 260.18 | 0.0010798 | 0.5085 | 589.2 | 2144 | 2733 |
| 150 | 374.78 | 0.0010905 | 0.39250 | 632.3 | 2114 | 2746 |
Different Units: Gauge Pressure and Absolute Pressure
Saturated steam tables can also use two different types of pressure: absolute pressure and gauge pressure.
- Absolute pressure is zero-referenced against a perfect vacuum.
- Gauge pressure is zero-referenced against atmospheric pressure (101.3 kPa, or 14.7 psi).
Saturated Steam Table using Absolute Pressure
| Press (Abs.) |
Temp. | Specific Volume | Specific Enthalpy | |||
|---|---|---|---|---|---|---|
| kPa | °C | m3/kg | kJ/kg | |||
| P | T | Vf | Vg | Hf | Hfg | Hg |
| 0 | -- | -- | -- | -- | -- | -- |
| 20 | 60.06 | 0.0010103 | 7.648 | 251.4 | 2358 | 2609 |
| 50 | 81.32 | 0.0010299 | 3.240 | 340.5 | 2305 | 2645 |
| 100 | 99.61 | 0.0010432 | 1.694 | 417.4 | 2258 | 2675 |
Saturated Steam Table using Gauge Pressure
| Press. (Gauge) |
Temp. | Specific Volume | Specific Enthalpy | |||
|---|---|---|---|---|---|---|
| kPaG | °C | m3/kg | kJ/kg | |||
| P | T | Vf | Vg | Hf | Hfg | Hg |
| 0 | 99.97 | 0.0010434 | 1.673 | 419.0 | 2257 | 2676 |
| 20 | 105.10 | 0.0010475 | 1.414 | 440.6 | 2243 | 2684 |
| 50 | 111.61 | 0.0010529 | 1.150 | 468.2 | 2225 | 2694 |
| 100 | 120.42 | 0.0010607 | 0.8803 | 505.6 | 2201 | 2707 |
Gauge pressure was created because it is often easier to reference measured pressure against the pressure we normally experience.
Steam tables based on gauge pressure indicate atmospheric pressure as 0, while steam tables based on absolute pressure indicate it as 101.3 kPa (14.7 psi). Also, to distinguish gauge pressure from absolute pressure, a "g" is typically added to the end of the pressure unit, for example kPaG or psig.
| Converting Gauge Units to Absolute Units |
|---|
For SI UnitsSteam Pressure [kPa abs] = Steam Pressure [kPaG] + 101.3 kPa For Imperial UnitsSteam Pressure [psi abs] = Steam Pressure [psiG] + 14.7 psi |
Important note: Problems can easily occur when absolute pressure is mistaken for gauge pressure (or vice versa), so it is always extremely important to pay close attention to the pressure units used in the table.
| Summary Table |
|---|
Gauge pressure:
Absolute pressure:
|
*Atmospheric pressure is 101.3 kPa (14.7 psi)
Superheated Steam Tables
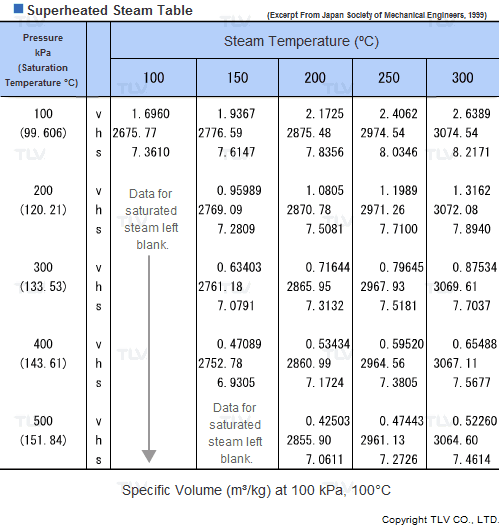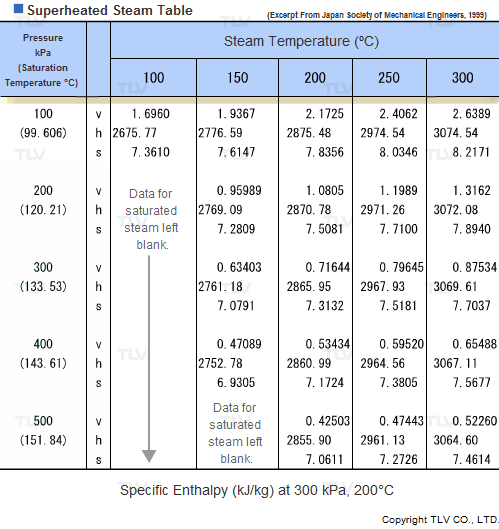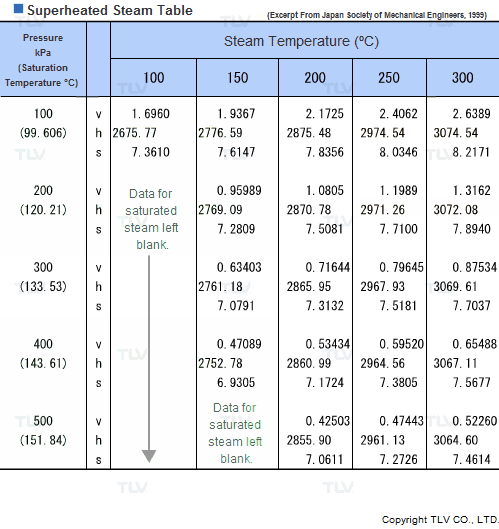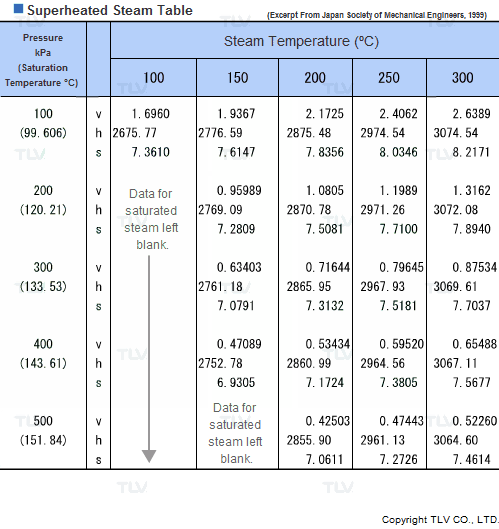
Values related to superheated steam cannot be obtained through a regular saturated steam table, but rather require the use of a Superheated Steam Table. This is because the temperature of superheated steam, unlike saturated steam, can vary considerably for a same pressure.
In fact, the number of possible temperature-pressure combinations is so great that it would be virtually impossible to gather them all in a single table. As a result, a large number of superheated steam tables use representative pressure-temperature values to form a summary table.
| Example of Superheated Steam Table |
|---|
|
|
| The above superheated steam table contains data about Specific Volume (Vg), Specific Enthalpy (Hg) and Specific Heat (Sg) at typical values of pressure and temperature. |
Related Posts

-
04/02/2023
1.What is Steam?
Steam is the gas formed when water passes from the liquid to the gaseous state. At the molecular level, this is when H2O molecules manage to break free from the bonds (i.e. hydrogen bonds) keeping them together.

-
04/02/2023
2.Principal Applications for Steam
Steam is used in a wide range of industries. Common applications for steam are, for example, steam heated processes in plants and factories and steam driven turbines in electric power plants, but the uses of steam in industry extend far beyond this. Here are some typical applications for steam in industry: Heating/Sterilization Propulsion/Drive Motive Atomization Cleaning Moisturization Humidification In the sections that follow, we will discuss various types of applications for steam, and provide some examples of steam-using equipment to illustrate them.

-
04/02/2023
3.Types of Steam
If water is heated beyond the boiling point, it vaporizes into steam, or water in the gaseous state. However, not all steam is the same. The properties of steam vary greatly depending on the pressure and temperature to which it is subject. In the article Principal Applications for Steam, we discussed several applications in which steam is used. In the sections that follow, we will discuss the types of steam used in these applications.

-
04/02/2023
4.Clean & Pure Steam
Have you considered the quality of your steam? In the production of certain goods such as food, electronics, and pharmaceutical products, a higher degree of steam quality is vital. In order to meet these needs, it would be ideal to use steam that is devoid of condensate, debris, and any other impurities (or almost so). The goal in producing filtered, clean, or pure steam is to get as close to the ideal state for each specific application as possible.

-
04/02/2023
5.Heating with Steam
Steam is one of the most common and effective heat transfer mediums used in industry, but it is not the only medium available. Other fluids such as hot water and oil are also used for indirect heating in heat exchangers. The following series of articles will focus on the advantages of using steam compared to hot water or oil for heating.

-
04/02/2023
6.Overall Heat Transfer Coefficient
The overall heat transfer coefficient, or U-value, refers to how well heat is conducted through over a series of resistant mediums. Its units are the W/(m2°C) [Btu/(hr-ft2°F)].
Expansion Joint Tozen Model JBF
Contact
Expansion Joint Tozen Model FJT
Contact
Flexible Joint Tozen SF SERIES
Contact
Copyright © 2017 Copyright by Phuc Minh Engineering Co., Ltd